There are several ways to purify salt water or dirty water. Distillation processes are most commonly used, as well as osmosis processes in which water passes through semipermeable membranes.
Distillation processes are based on heating water by heating with some fuel or sun, which produces water vapor which, when cooled, is converted into completely pure water.
In a similar way, nature turns seawater into rainwater. The sun heats water surfaces, water evaporates, and in clouds water vapor turns into pure water that falls to the ground in the form of drops.
This procedure can also be applied to the controlled process of obtaining pure water. For this, it is necessary to cover a certain surface of the water with a transparent cover in order to catch water vapor, and to cool that water and store it in clean water containers.
A device that can do this can be made in a very simple way.
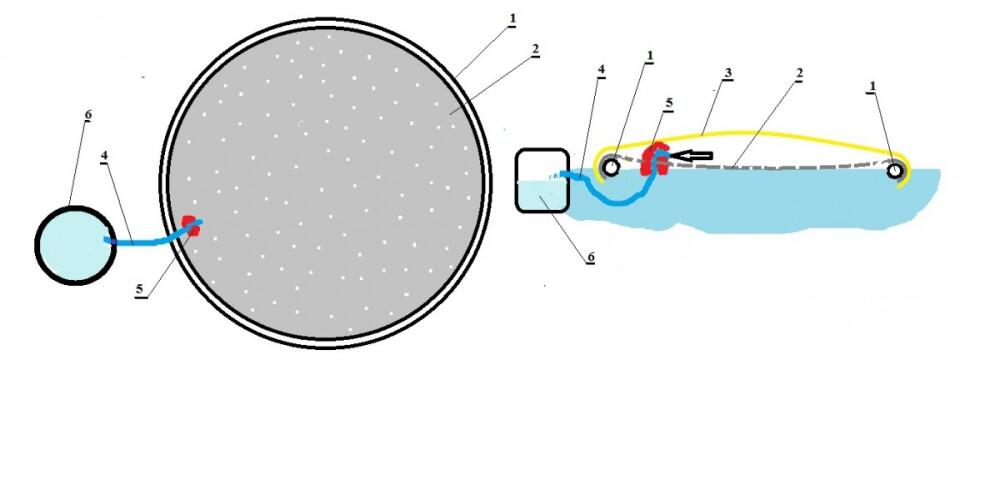
From the rubber hose (1) it is possible to make a large circle floating on the water surface. Two plastic foils are glued to this rubber hose (1) and their outer ends are immersed in the water on which they swim. The first is a dark perforated foil (2) that floats on the surface of the water, and above it is a solid transparent foil (3). Under the rubber hose (1) passes a water hose (4) which passes through a dark perforated foil (2), and the end of this water hose (4) is located under the transparent foil (3). The end of this water hose (4) is located above the water surface thanks to the float (5) which holds it above the water surface, and the very end of the water hose (4) rotates downwards so that in case of waves salt water cannot enter this water hose (4). The other end of the water hose (4) enters the clean water tank (6) where the produced drinking water is stored.
The process of water production is possible during sunny and warm weather and takes place under the action of the sun.
The sun’s rays fall on the dark perforated foil (2) and heat it up. This heat is transferred to the very surface of the water that is in contact with this foil. That way the surface of the water is much warmer than the water below. Even without this foil, the sun heats the water, but that heat is distributed to a greater depth, so the heating of the water is slower. The heated water surface evaporates much faster than it would without a dark perforated foil (2). The aerated water passes through small openings located on this perforated foil (2), and is retained under the transparent foil (3), and from there goes through the water hose (4). Upon entering this hose which is immersed in cold water the water vapor is cooled and converted back into running water. This running water flows into the clean water tank (6) from where the user can take it, and take it further.
Such a device can be placed on very large areas of the sea, or polluted lakes, swamps and wastewater lagoons. In order to prevent the rubber hose from moving during larger waves and strong winds, it would have to be anchored to the bottom.
This small portable device can be kept on sailboats or lifeboats.
In such a portable model, the rubber hose (1) would have to be made of a thin material and would be inflated before being placed on the surface of the water. The device would be kept in a coiled state in a box, and when needed, the rubber hose (1) would be inflated, so that both foils would spread over the surface of the water. One such device could produce enough drinking water for several people in an area of ten square meters in warm sunny weather. Before dark, or before bad weather, such a portable device could be repackaged by releasing air from the rubber hose (1) and placed in its box.
I have described most of my other innovations in this book.
Tags
Featured articles